Description
Mixtures of liquids can be separated according to their properties. Which technique to be used depends on a number of factors like miscibility or immiscibility of liquids. We use fractional distillation to separate the liquids based on their boiling points. Production of petroleum, diesel, tar are examples of fractional distillation. Initially, all these are part of crude oil.
The aim of this experiment is to demonstrate this concept.
Learning Outcomes
- A fun chemistry experiment for students where they learn about chromatography
- They learn about various separation techniques for liquids
- Correlation between theory and practical becomes easier
NEW LAUNCHES
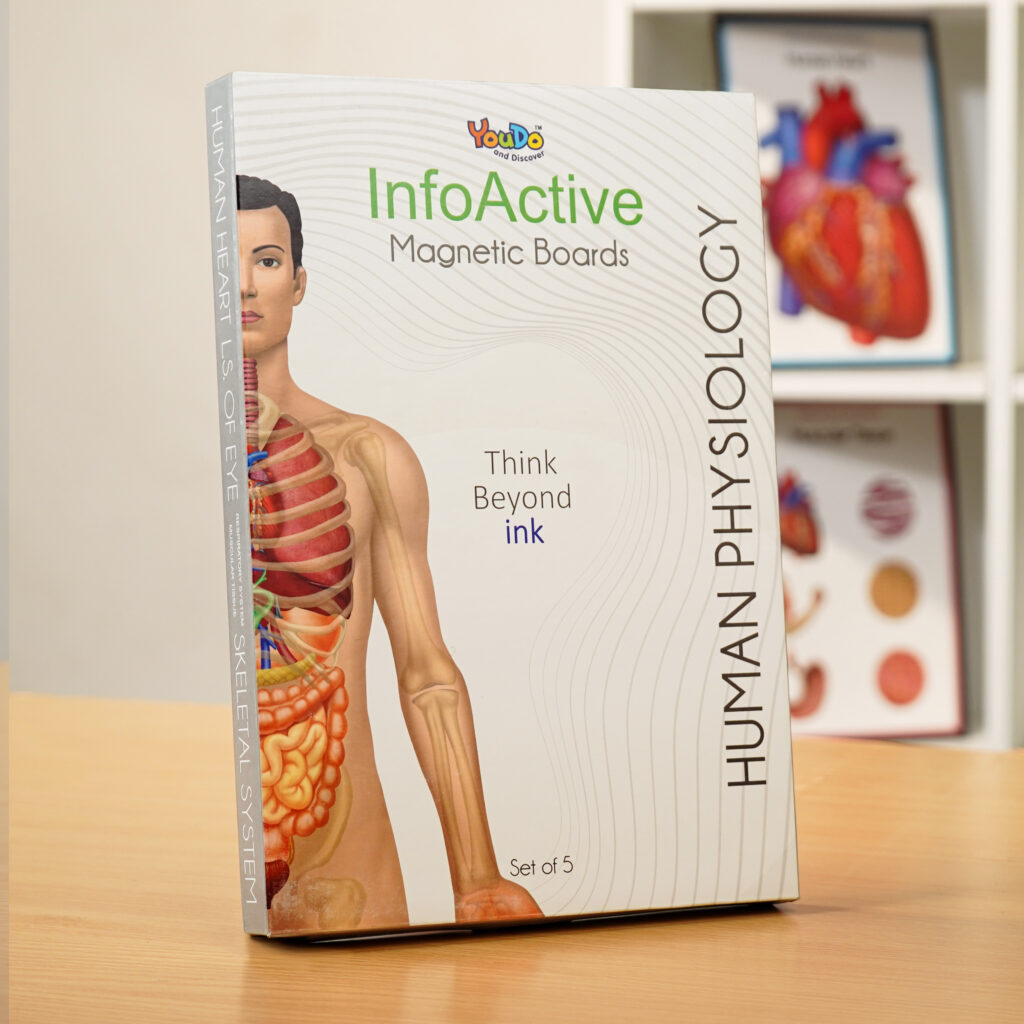
Info Magnetic Board
₹1999

Miniscope
₹699

Large Scribble Board
₹2999

Small Scribble Board
₹1999

Stick O Fun
₹1499

Hook N Look
₹647
We brainstorm and conceptualize.
Our designers brainstorm hundreds of ideas to create the most exciting projects that are also age-appropriate and educational.


Then prototype, test, iterate, test…
A variety of materials are tried out when creating prototypes. Every project goes through dozens of iterations!


Then test some more…with kids!
Our test crew of kids come join us every week to make sure that every project is engaging, fun, safe, and universally appealing.


Materials perfected
A variety of materials are tried out when creating prototypes. Every project goes through dozens of iterations!


then finally
Delivered to YOU!
Every month, you’ll receive a cool new project, with all the materials and instructions for hours of hands-on fun and learning.





















